
Consequently, the term cohort in clinical research is used to designate a group of subjects that have a characteristic, or a set of characteristics, in common (factor of study or exposure), and are followed over time. The word cohort means a group or group of people and has traditionally been associated with the military concept of the infantry corps of ancient Rome. The difference is that the researcher does not decide who is exposed, that is, does not assign the subject to one group or another the patients go to one group to another for reasons of routine or daily clinical practice. Ĭohort studies are similar to experimental studies since they are compared, exposed, and unexposed. Therefore, regardless of the type of research performed or evaluated in the clinical context, there must be appropriate tools to discriminate the best available evidence for health decision making. It should be noted that although clinical trials are considered the gold standard of clinical studies and are at the top of the traditional pyramid of scientific evidence, there may be limitations, for example, external validity that favors designs such as cohort studies. Only with a greater willingness to analyze these databases would it be possible to achieve a realistic understanding of how observational studies can best be used. suggested that observational studies usually provide valid information and could be used to explore the available databases. Therefore, observational studies are mainly used to identify risk factors and prognostic indicators, and in situations where randomized controlled trials would be impossible or unethical. 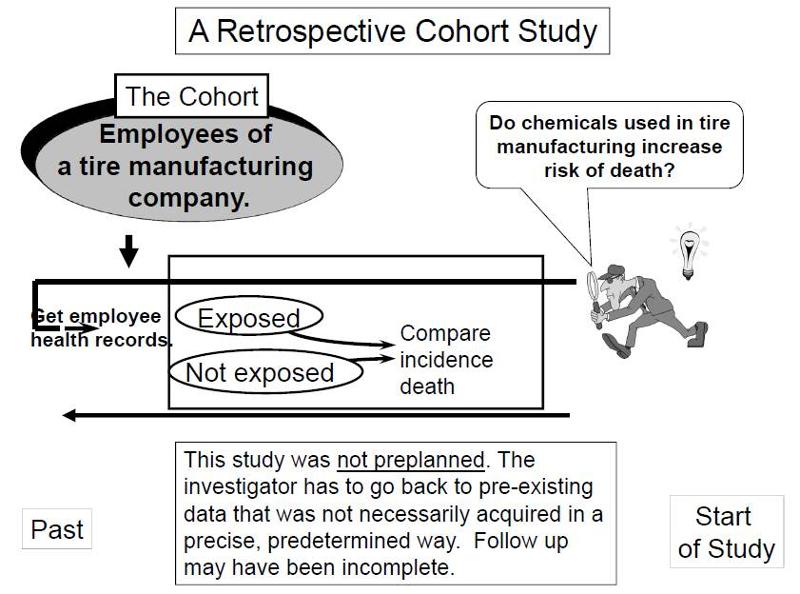
However, concerns about the inherent bias in these studies have limited their use when comparing treatments. In cohort studies, the design is similar to that of clinical trials, considered the most appropriate for causal inference, with the difference that exposure occurs naturally and is determined by preferences, clinical decisions or other conditions.Īs previously shown, cohorts, as well as other observational studies, have several advantages over randomized and controlled trials, including a lower cost, greater opportunity, and a wider range of patients. When a comparison group is provided, the study is defined as analytical, otherwise it is considered a description. In this context, observational studies can be classified according to the presence of a comparison group. This observational approach constitutes the typical environment of most clinical studies. Therefore, researchers cannot assign an exposure or treatment, but only observe the results. Therefore, health professionals can usually only observe situations and phenomena which are already segregated in groups. However, in daily clinical practice, experimental studies are difficult to carry out and often impose enormous logistical and budgetary challenges that are not easy to face. These results can be compared with those obtained by a different treatment. In general, biomedical research consists of two main categories: in an experimental approach, the researcher deliberately exposes the subjects to a specific treatment or intervention and observes the results. Finally, epidemiological research in public health and health services studies the frequency, distribution, and the health needs of the population, their risk factors, and their impact on public health. Clinical research studies the prevention, diagnosis, and treatment of diseases along with the knowledge of their natural history that can be categorized by the period of data collection (prospective, retrospective, and transversal) as well as by its design (observational or experimental), each with its own strengths and weaknesses. The basic or preclinical research seeks a better knowledge of the molecular, biochemical, and cellular mechanisms involved in the etiopathogenesis of diseases, forming the basis on which future studies are constructed. Approaches to research in health sciences.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
